|
The sodium ions are as close to the chloride ions as possible by fitting into the spaces between them. Compared to sodium ions, chloride ions are significantly larger. Lattice energies cannot be measured directly, but they can be estimated fairly well from the energies of the other processes described in the table immediately above. Six chloride ions with opposing charges surround each sodium ion in sodium chloride. It turns out that it is the lattice energy that renders the gift of stability to all ionic solids.Note that this lattice energy, while due principally to coulombic attraction between each ion and its eight nearest neighbors, is really the sum of all the interactions with the crystal. Thus, it requires 769 kJ to separate one mole of solid NaCl into gaseous Na + and Cl. 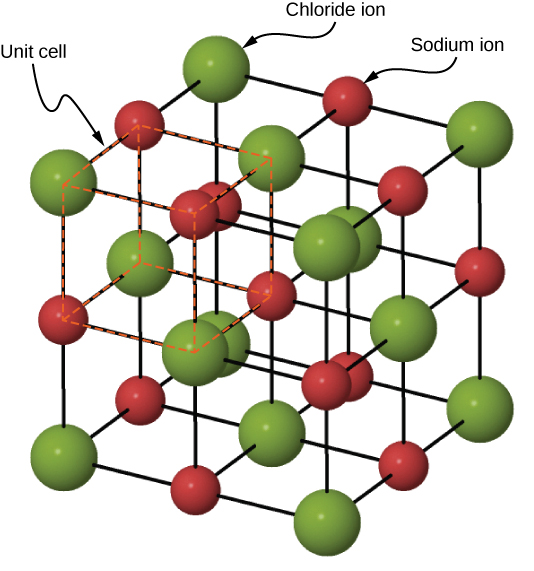
Its large magnitude should be no surprise, given the strength of the coulombic force between ions of opposite charge. Ionic solids exhibit a crystalline structure and tend to be rigid and brittle they also tend to have high melting and boiling points, which suggests that ionic bonds are very strong. The 787 kj/mol noted there is known as the NaCl lattice energy. A three-dimensional space-filling model for the ionic lattice in sodium chloride Remember that the lattice arrangement is giant - for example, a single grain of salt may contain 1.2 × 10 18. 
They cannot conduct electricity when they are solid because the ions are not free to move around and carry an electrical charge.Constitutes the big payoff in energy. Sodium Chloride (NaCl) is an ionic compound arranged in an endlessly repeating giant three-dimensional lattice structure. In this article, we will discuss sodium chloride (NaCl) lewis dot structure, crystal or lattice structure, is it polar or nonpolar, shape, unit cell, etc. Ionic compounds such as sodium chloride can only conduct electricity when they are melted or dissolved in water. In its aqueous form, it is called a saline solution. About 1 to 5 of seawater is made of NaCl. For the stable ionic crystalline structures, there is definite radius. Sodium chloride is an ionic compound with the chemical formula NaCl. The unit cell are described as simple cubic face centred and body centred unit cell. The smallest repeating part in the lattice is known as unit cell. This is the case with sodium chloride which has a melting point of 801☌. The crystalline solids have definite orderly arrangement of their constituent particles in three dimensions known as lattice. It takes a large amount of energy to overcome the strong electrostatic forces of attraction in an ionic compound and therefore they often have high melting and boiling points. 
(The arrangement of sulfide ions is identical to the arrangement of chloride ions in sodium chloride.) The radius of a zinc ion is only about 40 of the radius of a sulfide ion, so these small Zn 2 + ions are located in alternating tetrahedral holes, that is, in one. These forces act in all directions of the lattice and form ionic bonds. This structure contains sulfide ions on the lattice points of an FCC lattice. There are strong electrostatic forces of attraction that hold the oppositely charged ions together in the giant lattice. Ionic compounds such as sodium chloride are arranged in a giant three-dimensional lattice structure, which is a regular repeating pattern of positive and negative ions. This formula merely indicates that sodium chloride is made of an equal number of sodium and chloride ions. It is made of sodium ions which have lost an electron to become positively charged (Na+) and chloride ions which have gained an electron to become negatively charged (Cl-). crystal structure and is called a formula unit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
